 
The last major changes to the periodic table resulted from Glenn Seaborg's work in the middle of the 20th Century. With the discovery of isotopes of the elements, it became apparent that atomic weight was not the significant player in the periodic law as Mendeleev, Meyers and others had proposed, but rather, the properties of the elements varied periodically with atomic number. This charge, later termed the atomic number, could be used to number the elements within the periodic table. van den Broek in a series of two papers proposed that the atomic weight of an element was approximately equal to the charge on an atom. In the popular imagination the periodic system invariably and justifiably connects to his name, to the same extent that the theory of evolution connects to Darwin’s name and the theory of relativity to Einstein’s.Īlthough Mendeleev's table demonstrated the periodic nature of the elements, it remained for the discoveries of scientists of the 20th Century to explain why the properties of the elements recur periodically. Mendeleev’s version of the periodic table left the biggest impact on the scientific community, both at the time it was produced and thereafter. However, there was an untimely delay in the publication of his most elaborate periodic table, and, perhaps more importantly, Meyer-unlike Mendeleev-hesitated to make predictions about unknown elements. The closest precursor to Mendeleev’s table in both chronological and philosophical terms was developed by Julius Lothar Meyer, a German chemist, in 1864. By the 1860s a number of scientists had moved beyond the triad concept to produce some very respectable periodic systems. The elements in these groupings displayed an important numerical relationship to each other: the equivalent weight (an early substitute for atomic weight) of the middle element had the approximate mean of the values of the two flanking elements. The observation that certain types of elements prefer to combine with certain other types prompted early chemists to classify the elements in tables of chemical affinities. Mendeleev was hardly the first to arrive at a periodic system. It marks the 150th anniversary of the publication by the Russian chemist Dmitry Mendeleev (1834–1907) of his Periodic Table and celebrates the significance and impact of this outstandingly successful chart of the atomic building blocks of matter.  
The year of 2019 was designated by the United Nations as the International Year of the Periodic Table of Chemical Elements. This periodic table chart lists elements by name in alphabetical order including the element symbol and atomic number for quick and simple reference. Where #M# is the molar mass in grams per mole ( #gmol^-1#), #m# is the mass of the sample in grams ( #g#), and #n# is the amount in moles of the sample ( #mol#).Periodic Table with Element Names periodic table in alphabetical order by symbol Ask your teacher or tutor if you are not sure which one to use.Īnother way to find molar mass if amount and mass of a substance are known is by using the formula 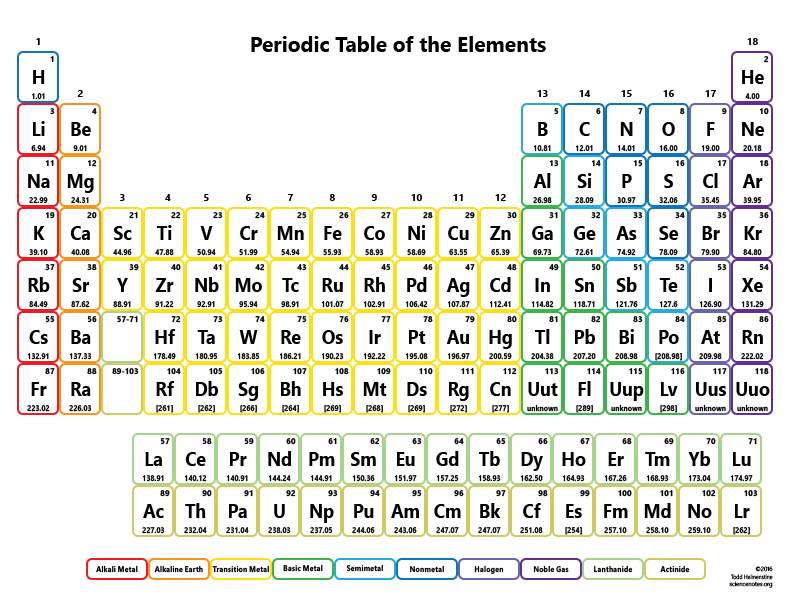
The molar masses will be slightly different if you are using a table with more accurate decimal values for atomic mass. Sulfuric acid #H_"2"SO_4# has 2 Hydrogen atoms (mass #1#), 1 sulfur atom (mass #32#) and 4 oxygen atoms (mass #16#) so: Then simply find out how many of each atom is in the molecule and add the respective atomic masses to find the molecular or molar mass.įor example water #H_2O# has 2 Hydrogen atoms (mass #1#) and 1 oxygen atom (mass #16#) so: I prefer to use tables with whole number atomic masses, but that is just personal preference. Find a periodic table and look at the atomic mass of each atom in the molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
